Verathon Expands Urology Portfolio with CFlex™, Its First Single-Use Flexible Cystoscope
Designed to simplify setup, improve visualization, and expand maneuverability in cystoscopy procedures
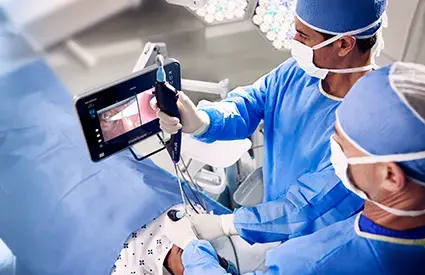
BOTHELL, Wash., May 15, 2026 — Verathon®, a global medical device company known for its GlideScope®, BFlex®, and BladderScan® brands, today announced the U.S. launch of CFlex™, an FDA 510(k)-cleared single-use flexible cystoscope. CFlex extends Verathon's urology portfolio into cystoscopy, applying the company's single-use endoscopy experience to a design built for cystoscopy workflow. The product will be featured at the Verathon Booth (#807) at AUA 2026 in Washington, D.C., May 15–18.
In both office and operating room settings, cystoscopy can be slowed by setup complexity, reduced irrigation flow during instrument use, and visualization challenges that extend procedure time. CFlex is engineered to address these challenges with integrated adapters, up to 70% more flow during instrument use1 compared with other currently available single-use cystoscopes, and high-contrast imaging2. Additionally, it features a tapered, slim 15 Fr insertion tube designed to support patient comfort.
What CFlex delivers for urologists:
- Reduces setup steps and supports clinician control of flow with a dedicated irrigation inlet, built-in adapters, and fingertip flow control, with fewer hand-offs during setup.
- Provides up to 70% more flow when an instrument is inserted1. Its larger (7.2 Fr) channel delivers more irrigation, helping maintain a clear field in cloudy or bloody environments.
- Supports clear visualization of anatomy with high-contrast imaging and accurate color2.
- Expands the field of view and supports access across the bladder with 210° bidirectional articulation, which can help reduce the need for repositioning.
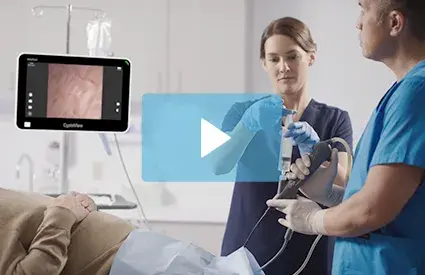
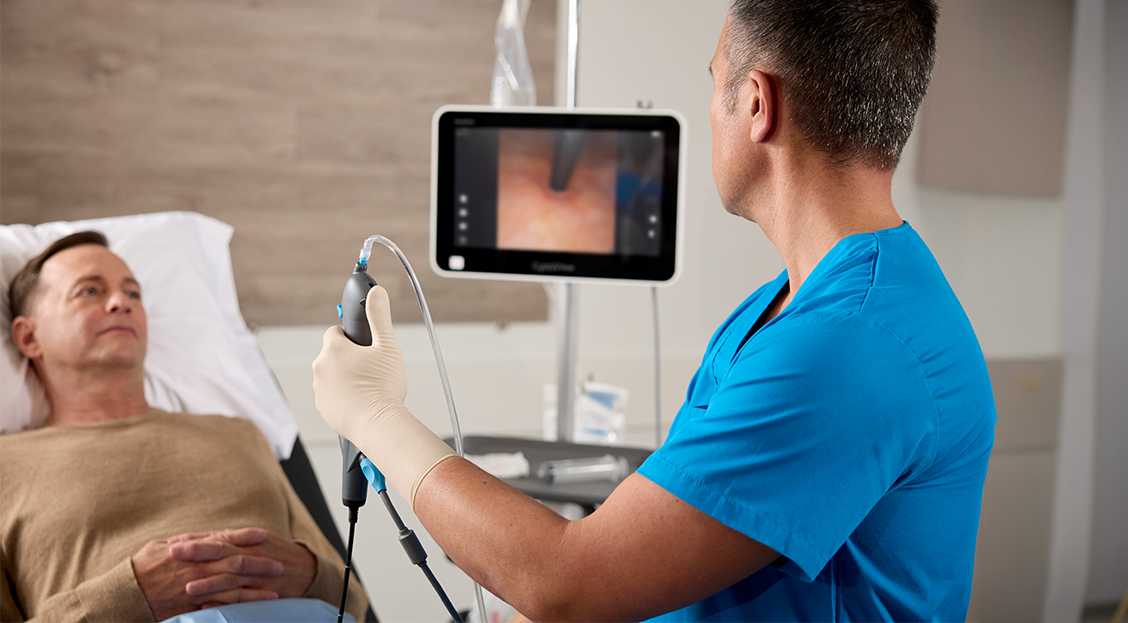
Beyond the scope itself, CFlex is designed for the full procedure; it pairs with Verathon's CystoView™ 15-inch Full HD monitor and a cystoscopy cart for quick setup across office and OR environments. Because every case starts with a new scope, CFlex eliminates reprocessing and scope repair cycles, removing a common source of case delays and reducing the risk of cross-contamination. CFlex also reflects Verathon's continued focus on sustainability, featuring a reusable cable to help reduce waste and a handle manufactured with 80% bio-based plastics calculated via the mass balance approach3.
Cystoscopy volumes in the U.S. are estimated to exceed two million procedures annually, with a growing share performed in office-based settings where setup speed and workflow simplicity are at a premium. CFlex is purpose-built for this shift.
"CFlex marks the next chapter for our urology business," said Parimal Shah, SVP, Urology Business Unit at Verathon. "We built a urology presence with BladderScan® and a single-use endoscopy platform with GlideScope® and BFlex®. CFlex brings those together, and it's the first of a broader urology portfolio."
Verathon will feature CFlex at Booth #807 during the 2026 American Urological Association Annual Meeting (AUA2026) at the Walter E. Washington Convention Center in Washington, D.C., May 15–18. Attendees can see live demonstrations, get hands-on previews, and meet with Verathon's urology team.
The CFlex Single-use Cystoscope is a sterile, single-use, flexible cystoscope intended for endoscopic access to and examination of the lower urinary tract in adult patients. CFlex is 510(k) cleared by the U.S. Food and Drug Administration and is now available in the United States. For more information or to request a demonstration, visit verathon.com/urology.
For More Information Contact:
Erna Kristinsdóttir
Senior Manager, Marketing Communications
1-800-331-2313
Erna.Kristinsdóttir@verathon.com
Visit the Verathon newsroom at https://www.verathon.com/news
1 Bench testing data on file. Comparison of flow rate during instrument use between CFlex™ and Ambu® aScope™ 4.
2 Bench testing data on file. Comparison of Ambu® aScope™ 4 Advance monitor and CystoView™.
3 Sourcing data on file supporting bio-based material content (mass balance approach).
Verathon® is a global medical device company dedicated to advancing single-use endoscopy and point-of-care visualization for airway management and urology. Its GlideScope® video laryngoscopes, BFlex® bronchoscopes, BladderScan® bladder volume systems, and CFlex™ single-use flexible cystoscopes are designed to help clinicians improve workflow efficiency and patient care. Headquartered in Bothell, Washington, and operating across North America, Europe, and Asia, Verathon is a business unit of Roper Technologies.