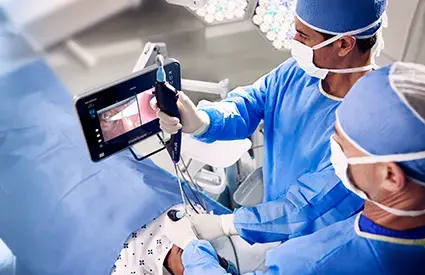
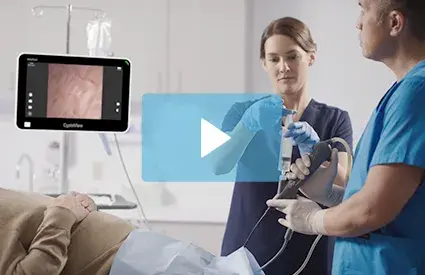
Recommendations and guidelines for the reprocessing of certain endoscopes types have changed in 2021 and 2022. Now the recently published Association for the Advancement of Medical Instrumentation (AAMI) standards have reclassified the reprocessing of flexible endoscopes as high-risk, and recommend additional steps when reprocessing these devices.
AAMI points to growing reports of multi-drug resistant organism (MDRO) transmission resulting in patient infection and high rates of mortality along with numerous healthcare-associated infections (HAIs) being linked to the use of contaminated endoscopes as to why the standards were updated. The new standards supports sterilizing all flexible endoscopes (semi-critical and critical) due to : 1)high microbial load after patient procedures, 2) complex design of flexible endoscopes, and 3)risk for biofilm formation.

Although endoscopes play a vital role in the effective delivery of health care and offer patients many benefits, the risks associated with iatrogenic transmission via endoscopes continue to be of significant concern. 1
Semi-critical devices come in contact with mucous membranes or non-intact skin. These include high-risk endoscopes such as duodenoscopes, bronchoscopes, ureteroscopes and cystoscopes.
AAMI recognizes the infection risk endoscopes can present to the patient and states: “It is advised that flexible and semi-rigid endoscopes to be used in semi-critical applications be sterilized prior to use.”
Acknowledging that transitioning from high level disinfection to sterilization can be a complex process, the committee encourages health care facilities partner with endoscope, reprocessor and sterilizer manufactures to help elevate the standard of endoscope processing.
This latest AAMI guidance comes after the 2021 FDA safety communication recommending the use of single-use endoscopes.
Now is the time to learn how single-use bronchoscopes can reduce the burden of reprocessing. Contact Verathon for more information.
1. National Standard. ANSI/AAMI ST91:2021. Flexible and semi-rigid endoscope processing in health care facilities.